SARS-CoV-2 Variant Identification Panel (RUO)

For Research Use Only. Not for use in diagnostic procedures.
Rapid and Cost-Effective Identification of SARS-CoV-2 Variants (Omicron Variant Covered)
The mutated SARS-CoV-2 has created a new challenge for labs and communities around the world. These concerned mutations increase COVID-19 transmissibility and possibly reduces the COVID-19 vaccine effectiveness. While next-generation sequencing-based assays are routinely used to track the new SARS-CoV-2 variants, they require sophisticated NGS lab equipment not accessible by most clinical laboratories, and the assays are expensive with a long turnaround time.
To provide a viable alternative, BGI has developed a rapid, sensitive and cost-effective solution with the assays run on PCR machines commonly found in most molecular diagnostic labs. These assays can easily be integrated into our current Emergency Use Authorization (EUA) workflow that includes high-throughput viral RNA extraction and SARS-CoV-2 detection for testing hundreds or thousands of samples a day.
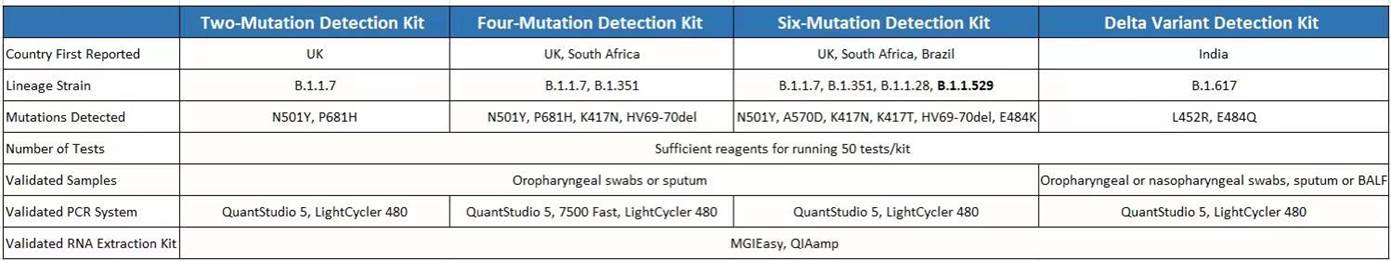
BGI is offering a suite of SARS-CoV-2 mutation detection kits that labs can purchase together with our EUA RT-PCR SARS-CoV-2 detection kit as a bundle to identify the SARS-CoV-2 and differentiate the lineage mutations. These mutation detection kits can be used independently without using the EUA detection kit.
Product Benefits
- Highly compatible – Bundled kits require very similar lab settings and procedure
- Highly sensitive –Superior limit of detection
- Fast TAT – Sample to result in 2.8 hours with automated sample preparation system (RT-PCR only takes 1 hour)
- High-throughput – Ramp up labs for large-scale, community-based testing
- Ease of use – All-inclusive with pre-mixed reaction reagents
- Easy interpretation – Analysis of each allele with well-defined controls (No bio-informatics required)
Related Information