HRD Detection Predicts Sensitivity to Platinum-based Chemotherapy for Ovarian Cancer patients in China - BGI Perspectives
2023-04-23
- First
prospective cohort study in China shows potential for more effective ovarian cancer
treatment
Homologous Recombination Deficiency (HRD) is a biomarker that predicts ovarian cancer treatment with PARP inhibitors or breast cancer treatment with first-line platinum-based chemotherapy. However, limited research is documented on platinum-based treatment prediction with HRD as a biomarker in ovarian cancer patients, especially in the Chinese population.
This first-ever China prospective cohort study, jointly conducted by BGI
Genomics clinical researcher Dr. Shao Di and the Fudan University Shanghai
Cancer Center team and published on the Journal
of Ovarian Research indicates that
HRD testing can accurately predict the sensitivity of platinum-based
chemotherapy for ovarian cancer patients.

The study results were published in the Journal of Ovarian Research
Research Method
The study covered 240 patients with primary treatment of high-grade serious ovarian cancer (HGSOC) admitted to the Cancer Hospital of Fudan University from January 2016 to September 2018 interval. Patients received more than two cycles of platinum-containing adjuvant chemotherapy after surgery and were classified as platinum-sensitive (Pt) and platinum-resistant according to the time of ovarian cancer recurrence.
All patients were tested with a gene panel test screening for 68 HRR genes, to assess the mutation status of HRR-related genes such as BRCA. Of these, 118 samples underwent the HRD test to assess loss of heterozygosity (LOH), telomere allele disequilibrium (TAI), and large segmental recombination abnormality (LST) status. The Kaplan-Meier method was used to plot survival curve and the effectiveness of these tests in predicting patient outcomes.
Results
Out of 240 HGSOC patients enrolled in the study, 82.5% had Pt-sensitive cancer with a platinum-free interval of more than six months. The study shows that 31.2% of patients had BRCA gene mutations, 53 in BRCA1 and 22 in BRCA2 (Figure 1). Additionally, 25% of patients had mutations in non-BRCA/HRR genes.
The
proportion of patients with positive HRD status was 64.4%. Such results reveal
that, compared to Western population, a higher proportion of Chinese ovarian
cancer patients benefit from PARP inhibitor maintenance therapy.
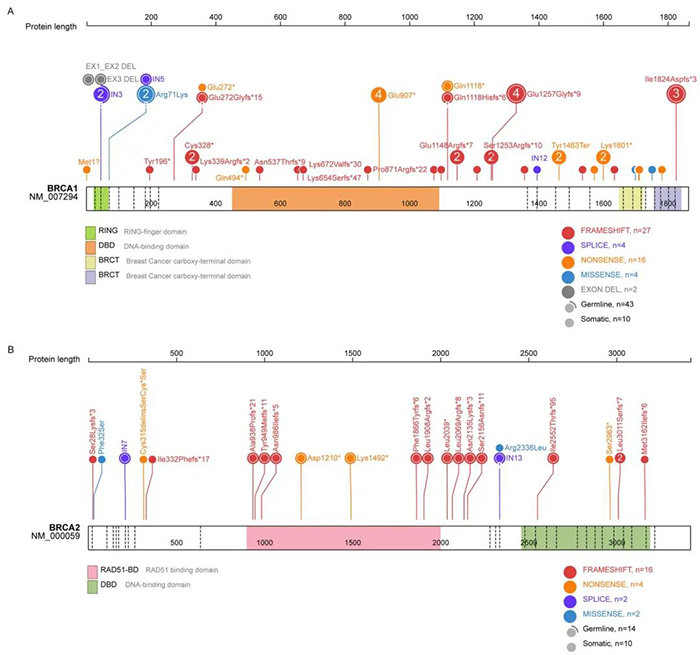
Figure 1: Distribution of BRCA1 and BRCA2 gene mutations. 31.2% of patients had BRCA gene mutations, 53 in BRCA1 and 22 in BRCA2
HRR gene mutation and HRD status are highly correlated with platinum chemotherapy sensitivity in patients with HGSOC.
Findings showed that the HRD score of platinum treatment-sensitive patients was slightly higher than that of Pt-resistant patients (Figure 2A).
Further analysis also found that the rate of Pt-sensitive patients in the HRD+ BRCAm group and HRD+ BRCAwt group was significantly higher than that in the HRD-BRCAwt group (Figure 2B).
Similarly, the study also found that Pt-sensitive patients were more enriched in the BRCA mutation group and non-BRCA HRR mutation group than HRR wt group.
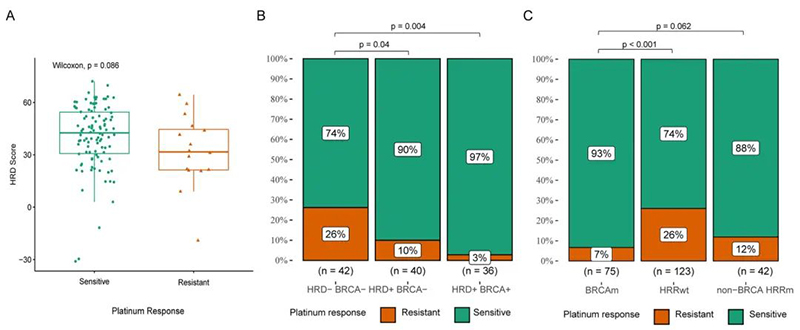
Figure 2: Correlation between HRD score, HRR mutation, HRD status and response to platinum chemotherapy
Analysis showed that patients with positive HRD status had a significantly longer progression-free survival (PFS) compared to those with negative HRD status (median PFS: 30.5 months vs 16.8 months, p=0.001 - Fig.3A)
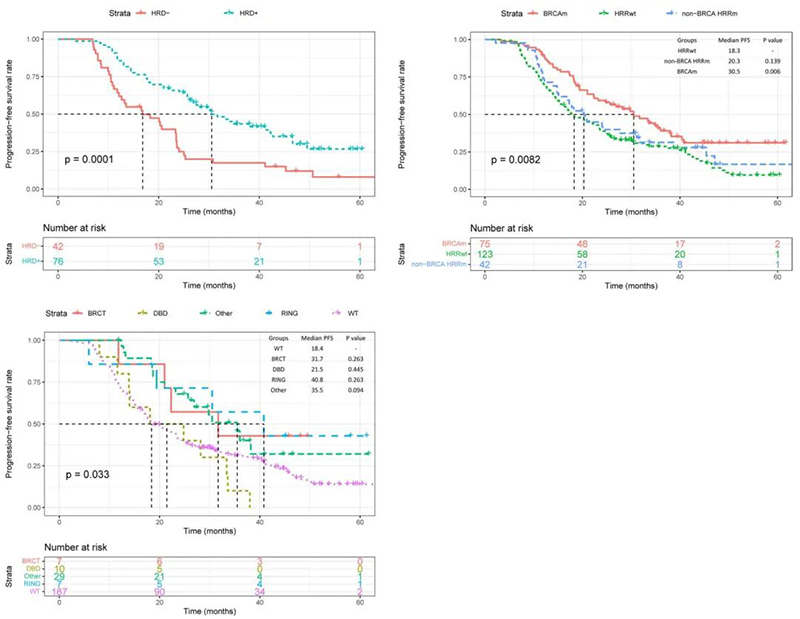
Figure 3: KM Survival Analysis Curves Grouped by HRD Status, HRR Gene Mutation, and BRCA1 Mutation Domain
Conclusion
This study demonstrated for the first time that HRD is a valid biomarker tool for predicting sensitivity to platinum-based chemotherapy in prospective cohort unselected Chinese ovarian cancer patients.
The study also found a higher rate of HRD positivity in Chinese ovarian cancer patients than in Western populations, suggesting that HRD testing could screen a higher proportion of Chinese PARP inhibitor population to facilitate precision therapy.
About BGI Genomics
BGI Genomics, headquartered in Shenzhen China, is the world's leading integrated solutions provider of precision medicine. Our services cover over 100 countries and regions, involving more than 2,300 medical institutions. In July 2017, as a subsidiary of BGI Group, BGI Genomics (300676.SZ) was officially listed on the Shenzhen Stock Exchange.